AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Cytotec and amniotic fluid embolism9/25/2023  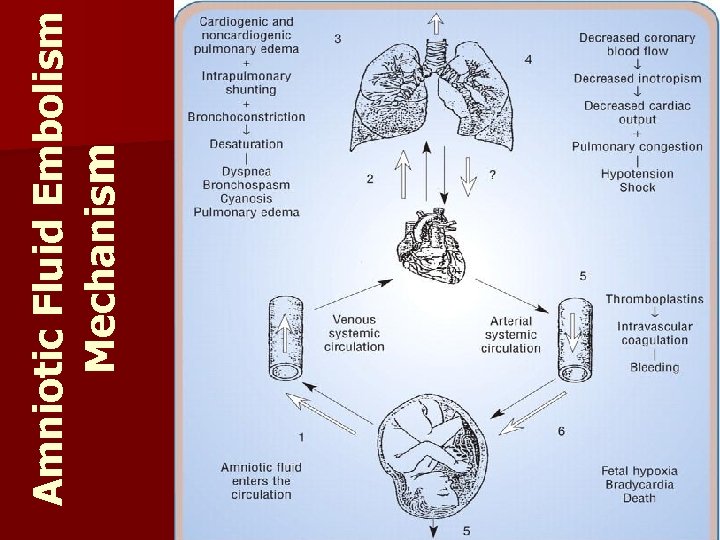 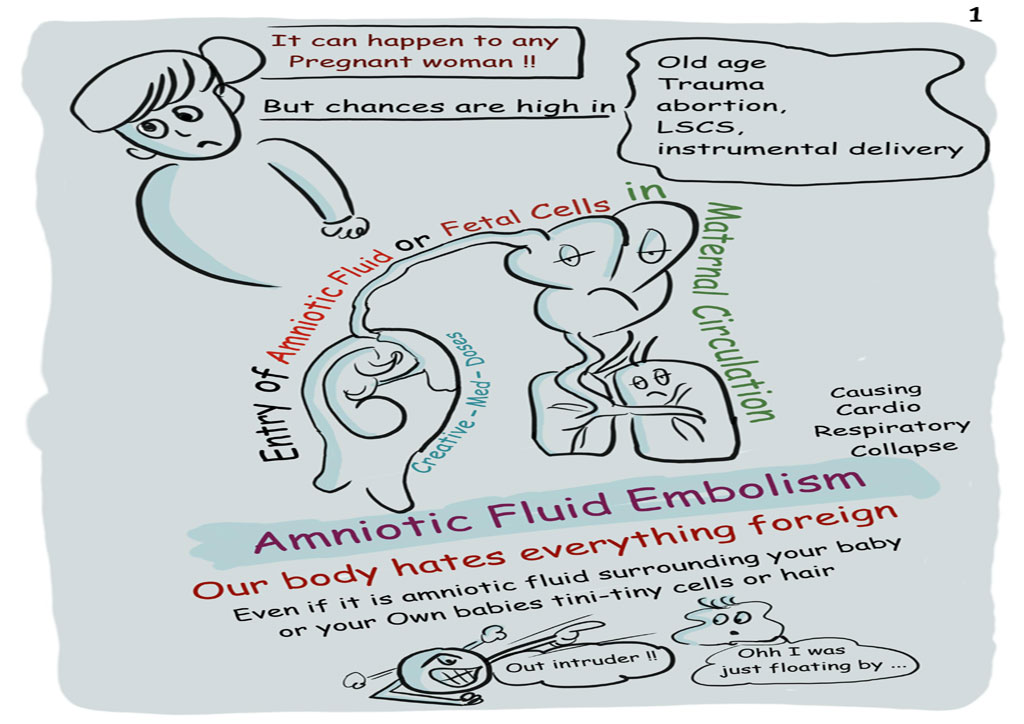
kills the drug if it can't be used by primary care providers" (Stolberg, The New York Times on-line edition, June 8). Eric Schaff, a pediatrician involved in clinical trials, explained: "The whole idea of was to increase access. As one columnist puts it, once RU-486 is approved, "this country can start putting this awful abortion fight behind us" (Mann, "We Need the Abortion Pill Now," The Washington Post, June 23, C9). They support RU-486 for its potential "to end the abortion debate" forever. As their comments reveal, they are not well-informed, or women's health is far from their primary concern. Shock, outrage and charges of betrayal characterize editorials lambasting the proposed restrictions. Many, especially in the media, do not share this viewpoint. While one can never condone using RU-486 to kill a child, the fact that the FDA may intend to contain the potential of this deadly drug for serious harm to women is a positive development. Given the serious risks the RU-486 regimen poses for women-from incomplete abortion, infection and excessive bleeding-the regulations are eminently reasonable. In addition, prescribing abortionists would be required to comply with the following: register with the drug's distributor be trained in RU-486 use (and so certified by a third party) be trained and certified in reading ultrasound scans to determine gestational age and to rule out life-threatening ectopic pregnancy and maintain admitting privileges at hospitals with emergency facilities no more than one hour from their office. Only licensed doctors who perform abortions will be allowed to prescribe RU-486. But in June Danco leaked new proposed restrictions on RU-486, hoping to raise a public outcry. Prior to June 2000, the FDA appeared eager to grant final approval. It's literally a mom and pop store" (quoted by Sharon Lerner, Village Voice, Aug. Its medical director, abortionist Richard Hausknecht, admits " is tiny. Much of its funding comes from foundations actively supporting population control, such as those set up by Warren Buffet, David Packard and George Soros. "There are no signs in the building's lobby or hallway pointing to the suite the company occupies." The phone is answered without mentioning Danco's name. The Wall Street Journal article also shines light into the secretive Danco Laboratories operation. Zimmerman reports that a "second, back-up manufacturing site is in the works, though its location couldn't be established, and the documents say it is having production problems, including 'poor yields' and 'high impurities'" (WSJ, id. government excoriates China for egregious violations of copyrights and patents on CDs and videos, but stands poised to permit importation of a pill-made and sold in violation of patent rights-in order to kill America's children. What exquisite irony this presents! The U.S. Zimmerman errs in a minor detail: Chinese plants are producing not RU-486 but a reverse-engineered copy of the pill, China being somewhat lax in observing intellectual property rights. The Population Council's manufacturing licensee, Danco Laboratories, will pay $293,363 this year to "one of several Chinese manufacturing plants that currently produce RU-486" (Zimmerman,"Awaiting Green Light, Abortion-Pill Venture Keeps to the Shadows," Wall Street Journal, Sept. 
It appears major pharmaceutical companies fear costly product-liability litigation as well as boycotts.Īs the September 30 deadline for FDA action looms, a manufacturer willing to supply mifepristone to U.S. Ulmann, former director of RU-486 development at Roussel-Uclaf, cites the "lack of a raw material manufacturer" as the main reason for the delay. Wanted: Manufacturer – Scruples Not Requiredĭr. He actually "wrote to Hoechst asking the company to file a new drug application with the FDA (an unprecedented situation in the pharmaceutical industry!)" which to their everlasting credit, "Hoechst intransigently refused to do" (André Ulmann, in "The Development of Mifepristone: A Pharmaceutical Drama in Three Acts," Journal of the American Medical Women's Assn. And that wasn't the end of his labors to bring chemical abortion to the U.S. market, except for use in clinical trials.Īpproval looked like a slam-dunk on President Clinton's first day in office when he signed an Executive Order asking federal agencies, including the FDA, to expedite approval of the abortion drug. 
rights to RU-486 (mifepristone) and four years after the Food and Drug Administration ("FDA") declared the drug "approvable," RU-486 has still not entered the U.S. Six years after the Population Council received U.S. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
